High specificity toward hepatobiliary diseases
The concentration of urinary bile acid sulfate elevates specifically in hepatobiliary diseases.
Especially, cholestasis and severe liver diseases indicate high value of urinary bile acid sulfate.
UBAS levels in various hepatobiliary diseases※1
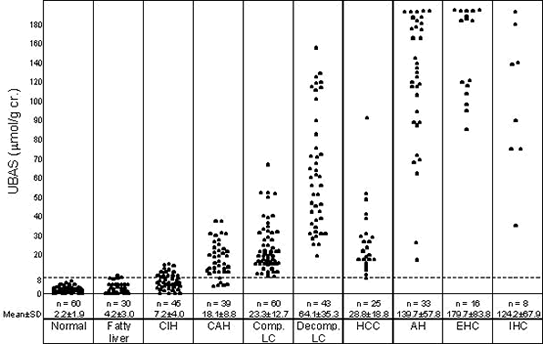
USBA levels is corrected by urinary creatinine levels. The concentration of bile acid sulfate in urine is then expressed as μmol/g creatinine.
| CIH: Chronic inactive hepatitis | CAH: Chronic active hepatitis |
| Comp. LC: Compensated liver cirrhosis | Decomp. LC:Decompensated liver cirrhosis |
| AH: acute hepatitis | EHC: Extrahepatic cholestasis |
| IHC: Intrahepatic cholestasis |
Determination of urinary bile acid sulfate UBASTEC-AUTO
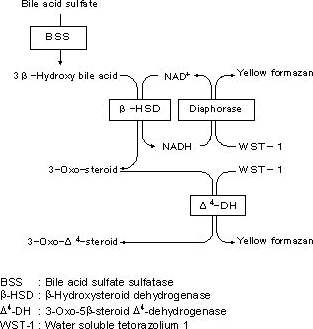
UBASTEC-AUTO is a simple assay kit for measurement of urinary total bile acid sulfate. UBASTEC-AUTO contains the enzyme, bile acid sulfate sulfatase which acts to the bile acid sulfate specifically.
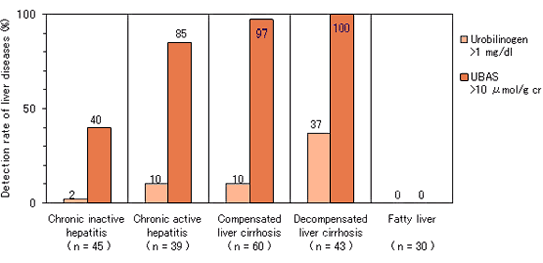
High detection rate of liver diseases in urine test.
In comparison with the urinary urobilinogen test, detection rate of liver diseases is markedly elevated by UBAS.
Comparison of detection rate of chronic liver diseases.※1
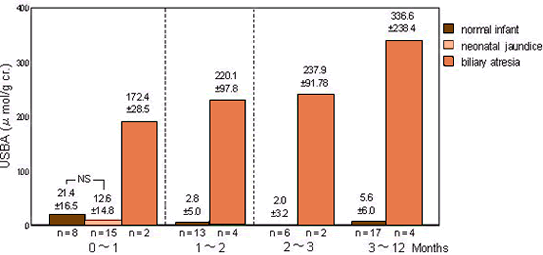
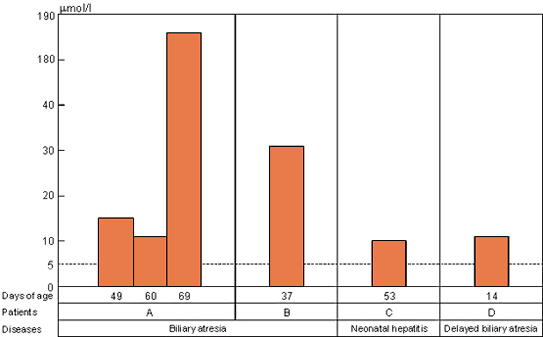
For screening of biliary atresia
UBAS is useful for screening of biliary atresia and for determining whether neonatal jaundice is caused by hepatobiliary diseases or not.
USBA levels in normal infant, neonatal jaundice and biliary atresia※1
Normal neonate value※6
Under 5 μmol/l (without creatinine corrected)
Screening of biliary atresia on neonatal urine.
| True negative | False positive | False negative | |
|---|---|---|---|
| Total (1,572 samples) | 97.9% (1,539 samples) |
2.1% (33 samples) |
0.0% (0 samples) |
| mmediately after birth (975 samples) |
98.5% (960 samples) |
1.5% (15 samples) |
0.0% (0 samples) |
| 1 month of age (597 samples) |
97.0% (579 samples) |
3.0% (18 samples) |
0.0% (0 samples) |
Biliary atresia was not included in total samples.
UBAS levels in neonatal liver diseases※6
Assay principal
Package and Method of storage
| Name | Constituent reagent | Package | Method of storage |
|---|---|---|---|
| UBASTEC-AUTO | Reagent 1 Enzyme | 22 ml use x 2 | Store protected from light at 4-8 ºC |
| Reagent 1 Solvent | 22 ml x 2 | ||
| Reagent 2 Enzyme | 6 ml use x 2 | ||
| Reagent 2 Solvent | 6 ml x 2 | ||
| Standard solution | 10 ml x 1 |
References
- ※1. Uenoyama R., Baba S., et al : KAN.TAN.SUI (Japanese), 31, 315-326 (1995)
- ※2. Meng G., Mashige F., Kitamura K., et al : Jpn. J. Clin. Chem., 23, 150-157 (1994)
- ※3. Adachi K., Kakigawa K., et al : Jpn. J. Clin. Chem., 26, 95-100 (1998)
- ※4. Matsui A., et al : The Journal of Pediatrics, 129, 306 (1996)
- ※5. Tazuke Y., Kodama N., Adachi K., et al : Jpn. J. Clin. Chem., 21, 249-258 (1992)
- ※6. Yamane H., et al : Kobe daigaku igakubu hokengakka kiyo (Japanese), 13, 1-8 (1997)